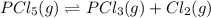Chemistry, 24.03.2020 21:15, elizzabel1944
Consider the system at equilibrium. PCl 5 ( g ) − ⇀ ↽ − PCl 3 ( g ) + Cl 2 ( g ) How will increasing the concentration of PCl5 shift the equilibrium? to the left to the right no effect How will increasing the concentration of PCl3 shift the equilibrium? to the right no effect to the left How will increasing the pressure by adding argon gas to the reaction mixture, while maintaining a constant volume, shift the equilibrium? to the right to the left no effect

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:30, robert7248
What is the percent composition of ca(oh)2? 37.7% ca, 53.0% o, and 10.3% h 45.5% ca, 38.2% o, and 16.3% h 54.0% ca, 43.0% o, and 2.7% h 64.7% ca, 27.0% o, and 8.3% h
Answers: 2

Chemistry, 22.06.2019 14:00, daniel1480
Which of the following is true about a carbonated soft drink? . the carbon dioxide is the solvent, and water is the solute.. the water is the solution, and carbon dioxide is the solvent.. the carbon dioxide is the solution, and the water is the solvent.. the water is the solvent, and the carbon dioxide is the solute.. .
Answers: 1

Chemistry, 22.06.2019 14:10, cameronbeaugh
13. a covalent bond between two atoms is likely to be polar if: a. one of the atoms is much more electronegative than the other. b. the two atoms are equally electronegative. c. the two atoms are of the same element. d. the bond is part of a tetrahedrally shaped molecule. e. one atom is an anion.
Answers: 1

Do you know the correct answer?
Consider the system at equilibrium. PCl 5 ( g ) − ⇀ ↽ − PCl 3 ( g ) + Cl 2 ( g ) How will increasing...
Questions in other subjects:


Biology, 01.08.2019 02:00

English, 01.08.2019 02:00


Mathematics, 01.08.2019 02:00


English, 01.08.2019 02:00

Biology, 01.08.2019 02:00

Physics, 01.08.2019 02:00


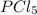 shift the equilibrium to the right side.
shift the equilibrium to the right side.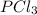 shift the equilibrium to the left.
shift the equilibrium to the left.