
Gaseous butane will react with gaseous oxygen to produce gaseous carbon dioxide and gaseous water . Suppose 35.5 g of butane is mixed with 33. g of oxygen. Calculate the minimum mass of butane that could be left over by the chemical reaction. Round your answer to significant digits.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:10, aleilyg2005
Select the correct answer from each drop-down menu. describe what happens to a carbon-11 atom when it undergoes positron emission. the decay of a carbon-11 atom _1_, and this causes it to emit _2_.options for 1: > changes a neutron into a proton> changes a proton into a neutron> is hit with a neutron> reconfigures its protons and neutronsoptions for 2: > a negatively charged electron-sized particle> a positively charged election-sized particle> two atoms and several neutrons> two neutrons and two protons
Answers: 3

Chemistry, 22.06.2019 13:00, nadiarose6345
In a copper wire, a temperature increase is the result of which of the following
Answers: 1


Chemistry, 22.06.2019 19:30, toriabrocks
If 16.00g of hydrogen gas reacts with 126.73g of oxygen, how many grams of water are yielded? (both reactants are completely consumed in the reaction.)
Answers: 2
Do you know the correct answer?
Gaseous butane will react with gaseous oxygen to produce gaseous carbon dioxide and gaseous water ....
Questions in other subjects:

Social Studies, 20.10.2019 11:50


Health, 20.10.2019 11:50

History, 20.10.2019 11:50

History, 20.10.2019 11:50



Health, 20.10.2019 11:50


Mathematics, 20.10.2019 11:50


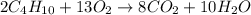
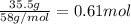
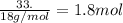
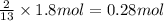 of butane
of butane



