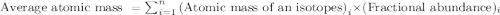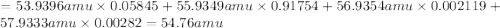Chemistry, 24.03.2020 17:02, ameliaxbowen65
Iron has many isotopes but only 4 are found in significant amounts in naturally found mixtures. The amounts by mass percent are: 5.845% of 54 Fe (53.9396 amu) 91.754% of 56Fe (55.9349 amu), 2.119% of 57Fe (56.9354 amu) and 0.282% of 58Fe (57.9333 amu). What would you determine the average mass of iron to be

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 23.06.2019 03:30, antoinetteee03
Name atleast 3 type of energy associated with the microwave
Answers: 1

Chemistry, 23.06.2019 15:00, WampWamp8751
For part 1, describe the changes in the colors of the well, if any, as you go from well 1 to well 9—that is, as you go from the well with the least copper(ii) nitrate to the well with the most copper(ii) nitrate. which wells had the most distinct precipitate? for part 2, describe the changes in the colors of the well, if any, as you go from well 1 to well 9—that is, as you go from the well with the least iron(ii) sulfate to the well with the most iron(ii) sulfate. which wells had the most distinct precipitate? for part 3, describe the changes in the colors of the well, if any, as you go from well 1 to well 9—that is, as you go from the well with the least iron(iii) nitrate to the well with the most iron(iii) nitrate. which wells had the most distinct precipitate?
Answers: 3

Chemistry, 23.06.2019 22:40, quintasiahaskin
The pressure on 7.0 l of gas is increased from 15 psi to 1420 torr, at constanttemperature. what is the new volume of the gas?
Answers: 2

Chemistry, 24.06.2019 00:00, elviaortiz402373
Percent purity of fe2o3 in original sample. fe2o3 + 3co - 2fe+3co2. 1.39 x 10^3 kg of fe is obtained from 2.22 x 10^3 kg sample of fe2o3. reaction goes to completion
Answers: 1
Do you know the correct answer?
Iron has many isotopes but only 4 are found in significant amounts in naturally found mixtures. The...
Questions in other subjects:



Mathematics, 26.06.2020 16:01

Mathematics, 26.06.2020 16:01



Mathematics, 26.06.2020 16:01