Chemistry, 24.03.2020 16:19, juanaaraujo1104
Base your answer on the information below and your knowledge of chemistry. Methanol can be manufactured by a reaction that is reversible. In the reaction, carbon monoxide gas and hydrogen gas react using a catalyst. The equation below represents this system at equilibrium. CO(g) 2H2(g) CH3OH(g) energy. Explain, in terms of collision theory, why increasing the concentration of H2(g) in this system will increase the concentration of CH3OH(g).

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:10, alanflores40
Amonoprotic acid is an acid that donates a single proton to the solution. suppose you have 0.140 g of a monoprotic acid dissolved in 35.0 ml of water. this solution is then neutralized with 14.5 ml of 0.110 m naoh. what is the molar mass of the acid?
Answers: 1


Chemistry, 22.06.2019 14:50, alexabbarker9781
How are evaporation and sublimation similar? a both involve the formation of a gas. b both release energy to the surroundings. c both take place throughout a solid. d both take place at the surface of a liquid.
Answers: 1

Chemistry, 22.06.2019 15:00, NatalieKnows
Areaction is first order with respect to reactant x and second order with respect to reactant y. which statement describes the rate law for this reaction?
Answers: 1
Do you know the correct answer?
Base your answer on the information below and your knowledge of chemistry. Methanol can be manufactu...
Questions in other subjects:

Chemistry, 02.04.2020 00:43







Mathematics, 02.04.2020 00:43


Chemistry, 02.04.2020 00:43


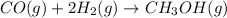
![Rate=k[CO]^x[H_2]^y](/tpl/images/0560/8875/426a0.png)
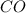 and
and