
Chemistry, 24.03.2020 07:15, StevencoolGG
A reaction vessel contains 2.50 moles of ci2 and 6.15 moles of f2 how many moles of excess reactant are left

Answers: 3
Other questions on the subject: Chemistry




Chemistry, 22.06.2019 10:00, Cythina2007
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1
Do you know the correct answer?
A reaction vessel contains 2.50 moles of ci2 and 6.15 moles of f2 how many moles of excess reactant...
Questions in other subjects:

Spanish, 09.09.2020 22:01


Mathematics, 09.09.2020 22:01

English, 09.09.2020 22:01

Mathematics, 09.09.2020 22:01



Mathematics, 09.09.2020 22:01

Mathematics, 09.09.2020 22:01


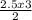 = 3.75moles of F₂
= 3.75moles of F₂



