
Chemistry, 24.03.2020 05:24, Alizerodriguez2010
In a laboratory activity, a student titrates a 20.0 milliliter sample of Hcl(aq) using 0.025 M NAOH (aq). In one of the titration trails, 17.6 milliliters of the base solution exactly neutralizes the acid sample. Calculate the concentration f the hydrochloric acid using the titration data

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 11:00, daniel1480
Problem page combustion of hydrocarbons such as pentane ( c5 h12 ) produces carbon dioxide, a "greenhouse gas." greenhouse gases in the earth's atmosphere can trap the sun's heat, raising the average temperature of the earth. for this reason there has been a great deal of international discussion about whether to regulate the production of carbon dioxide.(a) write a balanced chemical equation, including physical state symbols, for the combustion of liquid pentane into gaseous carbon dioxide and gaseous water. (b) suppose 0.350 kg of pentane are burned in air at a pressure of exactly 1 atm and a temperature of 20.0 degree c. calculate the volume of carbon dioxide gas that is produced. be sure your answer has the correct number of significant digits.
Answers: 2

Chemistry, 22.06.2019 12:30, hala201490
Place the elements below in order of decreasing ionization energy. aluminum(al) chlorine(cl) magnesium (mg) sulfur(s)
Answers: 1
Do you know the correct answer?
In a laboratory activity, a student titrates a 20.0 milliliter sample of Hcl(aq) using 0.025 M NAOH...
Questions in other subjects:

English, 03.06.2021 01:10




Engineering, 03.06.2021 01:10



Mathematics, 03.06.2021 01:10



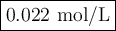

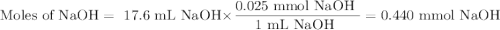
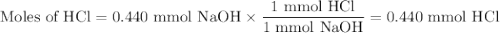
![\text{[HCl]} = \dfrac{\text{0.440 mmol HCl}}{\text{20.0 mL HCl}} = \textbf{0.022 mol/L}\\\text{The concentration of HCl is $\large \boxed{\textbf{0.022 mol/L}}$}](/tpl/images/0560/5762/e6247.png) }
}




