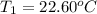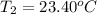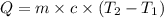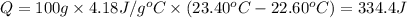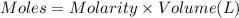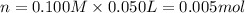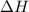Chemistry, 24.03.2020 00:29, PrincesssOfficial
In a coffe cup calorimeter, 50.0mL of 0.100M of AgNO3 and 50mL of 0.100M HCl are mixed to yield the following reaction:
Ag+ (aq) + Cl -==> AgCl(s)
The two solutions were initially at 22.60°C, and the final temperature is 23.40°C. How do I calculate the heat that accompanies this reaction in kJ/mol, assuming that the combined solution has a mass of 100g and a specific heat capacity of 4.18 J/g°C.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:20, kingsqueen883
Consider the two electron arrangements for neutral atoms a and b. are atoms a and b the same element? a - 1s2, 2s2, 2p6, 3s1 b - 1s2, 2s2, 2p6, 5s1
Answers: 3


Chemistry, 22.06.2019 19:30, Sumitco9578
Anurse used a 0.02-mg/l solution of disinfection to clean a patients wound. what is the concentration of the solution expressed as a percentage?
Answers: 1

Chemistry, 22.06.2019 21:00, itasykamila
How many neutrons does an element have if its atomic number is 50 and its mass number is 166
Answers: 1
Do you know the correct answer?
In a coffe cup calorimeter, 50.0mL of 0.100M of AgNO3 and 50mL of 0.100M HCl are mixed to yield the...
Questions in other subjects: