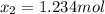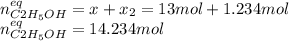Chemistry, 23.03.2020 23:20, little68941
While ethanol (CH3CH2OH) is produced naturally by fermentation, e. g. in beer- and wine-making, industrially it is synthesized by reacting ethylene (CH2CH2) with water vapor at elevated temperatures A chemical engineer studying this reaction fills a 75.0 L tank at 18. °C with 29. mol of ethylene gas and 16. mol of water vapor. He then raises the temperature considerably, and when the mixture has come to equilibrium determines that it contains 16. mol of ethylene gas and 3.0 mol of water vapor. 囲 The engineer then adds another 15. mol of ethylene, and allows the mixture to come to equilibrium again. Calculate the moles of ethanol after equilibrium is reached the second time. Round your answer to 2 significant digits.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 04:40, deedee363
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3

Chemistry, 22.06.2019 13:00, netflixacc0107
Amixture with the same composition throughout is!
Answers: 1
Do you know the correct answer?
While ethanol (CH3CH2OH) is produced naturally by fermentation, e. g. in beer- and wine-making, indu...
Questions in other subjects:


Mathematics, 28.03.2020 21:38

Mathematics, 28.03.2020 21:38

Mathematics, 28.03.2020 21:38



English, 28.03.2020 21:38

Mathematics, 28.03.2020 21:38



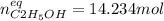
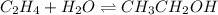
![Kc=\frac{[CH_3CH_2OH]_{eq}}{[H_2O]_{eq}[CH_2CH_2]_{eq}}](/tpl/images/0559/8704/4cbd4.png)
 result:
result:![[CH_2CH_2]_{eq}=29mol-x=16mol\\x=29-16=13mol](/tpl/images/0559/8704/ac34b.png)
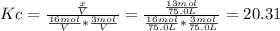
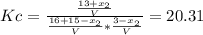
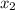 finally result (solving by solver or quadratic equation):
finally result (solving by solver or quadratic equation):