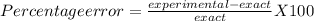Chemistry, 21.03.2020 22:38, imjustdumbsis
The exact value for the density of aluminum is 2.699 g. cm^3. Working in the science lab at school, Joseph finds the density of a piece of aluminum to be 2.75 g/cm^3. What is Joseph's percent error? (Round to the nearest hundredth.)

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:00, fgcherubin
Consider the balanced equation below. n2h4 + 2h2o2 n2 + 4h2o what are the mole ratios of hydrazine (n2h4) to hydrogen peroxide (h2o2) and hydrazine to water? 1: 2 and 1: 4 1: 3 and 1: 4 1: 2 and 3: 5 1: 3 and 3: 5
Answers: 3

Chemistry, 22.06.2019 10:00, berniceallonce22
What is the atomic mass of an atom that has 6 protons, 6 neutrons, and 6 electrons? a) 6 b) 8 c) + 1 d) 12 e) 18
Answers: 1


Chemistry, 22.06.2019 15:00, emmalie52
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1
Do you know the correct answer?
The exact value for the density of aluminum is 2.699 g. cm^3. Working in the science lab at school,...
Questions in other subjects:

Mathematics, 07.12.2021 08:50



English, 07.12.2021 08:50


Mathematics, 07.12.2021 08:50



English, 07.12.2021 08:50