
Chemistry, 20.03.2020 11:14, ciarrap552
If you assume this reaction is driven to completion because of the large excess of one ion, what is the concentration of [Fe(SCN)]2+ that would be formed from 6.00 mL of 0.00180 M KSCN 5.00 mL 0.240 M Fe(NO3)3 and 14.00 mL of 0.050 M HNO3.
Question 3 options:
0.240 M
4.32 x 10^-4
0.0480
0.0460 M

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:30, mimithurmond03
A6.10 m nacl can be made by adding [x]g of nacl to a container and making the volume of water up to the 1.00 l line
Answers: 1

Chemistry, 21.06.2019 22:30, 20alondra04
In order to calculate the amount of heat transferred you must know the __ and specific heat of the material, as well as the change in temperature. a. volume b. density c. mass d. enthalpy
Answers: 1


Chemistry, 22.06.2019 11:30, samantha9430
Determine the reaction and balance the following equations urgent due in the morning
Answers: 2
Do you know the correct answer?
If you assume this reaction is driven to completion because of the large excess of one ion, what is...
Questions in other subjects:





Mathematics, 15.07.2020 21:01



Mathematics, 15.07.2020 21:01



![[Fe(SCN)]^{2+}](/tpl/images/0556/0307/0c409.png) is,
is, 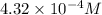
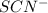 and
and 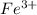 is excess reagent.
is excess reagent.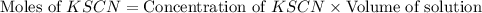
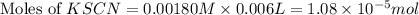
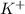 = Moles of
= Moles of 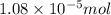
![\text{Concentration of }[Fe(SCN)]^{2+}=\frac{\text{Moles of }[Fe(SCN)]^{2+}}{\text{Volume of solution}}](/tpl/images/0556/0307/e16b4.png)
![\text{Concentration of }[Fe(SCN)]^{2+}=\frac{1.08\times 10^{-5}mol}{0.025L}=4.32\times 10^{-4}M](/tpl/images/0556/0307/cd669.png)





