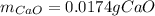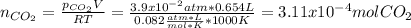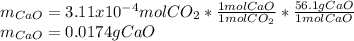Chemistry, 20.03.2020 11:04, Affousietta
A sample of CaCO3(s) is introduced into a sealed container of volume 0.654 L and heated to 1000 K until equilibrium is reached. The Kp for the reaction CaCO3(s) ∆ CaO(s) + CO2(g) is 3.9 * 10-2 at this temperature. Calculate the mass of CaO(s) that is present at equilibriu

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 20:00, jalenevoyles
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3

Chemistry, 22.06.2019 22:30, gonzalesalexiaouv1bg
What if it is did darwin used to support his theory of evolution
Answers: 1

Chemistry, 23.06.2019 07:30, 22emilyl530
Chris is about to do an experiment to measure the density of water at several temperatures. his teacher has him prepare and sign a safety contract before beginning the experiment. which term is mostlikely part of the safety contract
Answers: 3
Do you know the correct answer?
A sample of CaCO3(s) is introduced into a sealed container of volume 0.654 L and heated to 1000 K un...
Questions in other subjects:


Mathematics, 04.02.2020 14:46


Mathematics, 04.02.2020 14:46

English, 04.02.2020 14:46

Mathematics, 04.02.2020 14:46


Mathematics, 04.02.2020 14:46