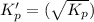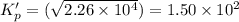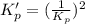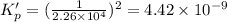Chemistry, 20.03.2020 09:47, jessicamcummins
This reaction has an equilibrium constant of Kp = 2.26 * 104 at 298 K: CO(g) + 2 H2(g) ∆ CH3OH(g) Calculate Kp for each reaction and predict whether reactants or products will be favored at equilibrium:

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 06:00, joelpimentel
This flow chart shows the amount of energy that is emitted by each type of light. ultraviolet > blue light > yellow light > red light (maximum energy) (minimum energy) in an experiment, shining which type of light on a strip of metal would be least likely to produce the photoelectric effect? ultraviolet light dim blue light bright red light bright yellow light
Answers: 2

Chemistry, 22.06.2019 10:40, rntaran2002
What is the ph of a 0.0010 m hno3? 1.0 3.0 4.0 5.0
Answers: 2
Do you know the correct answer?
This reaction has an equilibrium constant of Kp = 2.26 * 104 at 298 K: CO(g) + 2 H2(g) ∆ CH3OH(g) Ca...
Questions in other subjects:










Mathematics, 09.08.2021 21:10


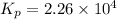 at 298K.
at 298K.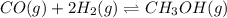
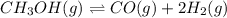
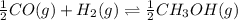
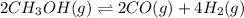
 : reactants are favoured
: reactants are favoured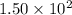 : products are favoured
: products are favoured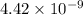 : reactants are favoured
: reactants are favoured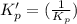
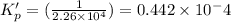
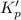 is less than 1, reactants will be favored at equilibrium.
is less than 1, reactants will be favored at equilibrium.