
Chemistry, 20.03.2020 08:30, darwin59651
Calculate the pH of a 0.437 M aqueous solution of hydrocyanic acid (HCN, Ka = 4.0×10-10) and the equilibrium concentrations of the weak acid and its conjugate base.

Answers: 2
Other questions on the subject: Chemistry




Chemistry, 22.06.2019 17:10, sophiaa23
)benzene and toluene form nearly ideal solutions. consider an equimolar solution of benzene and toluene. at 20 °c the vapour pressures of pure benzene and toluene are 9.9 kpa and 2.9 kpa, respectively. the solution is boiled by reducing the external pressure below the vapour pressure. calculate (i) the pressure when boiling begins, (ii) the composition of each component in the vapour, and (iii) the vapour pressure when only a few drops of liquid remain. assume that the rate of vaporization is low enough for the temperature to remain constant at 20 °c.
Answers: 1
Do you know the correct answer?
Calculate the pH of a 0.437 M aqueous solution of hydrocyanic acid (HCN, Ka = 4.0×10-10) and the equ...
Questions in other subjects:

Mathematics, 12.10.2020 22:01


History, 12.10.2020 22:01




Social Studies, 12.10.2020 22:01




![[HCN]_{eq}=0.43699M](/tpl/images/0555/6850/8a21f.png)
![[CN^-]_{eq}=1.322x10^{-5}M](/tpl/images/0555/6850/83c77.png)
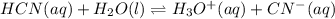
![Ka=\frac{[H^+]_{eq}[CN^-]_{eq}}{[HCN]_{eq}}](/tpl/images/0555/6850/4425b.png)
 due to the reaction extent, goes:
due to the reaction extent, goes: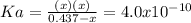
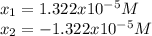
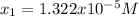
![pH=-log([H^+])=-log(1.322x10^{-5})=4.88](/tpl/images/0555/6850/f810b.png)
![[HCN]_{eq}=0.437M-1.322x10^{-5}M=0.43699M](/tpl/images/0555/6850/d4f3f.png)




