Chemistry, 20.03.2020 06:29, lisafrench8222
The equilibrium constant Kp for the reaction I2(g) + Br2(g) ⇀↽ 2 IBr(g) + 11.7 kJ is 280 at 150◦C. Suppose that a quantity of IBr is placed in a closed reaction vessel and the system is allowed to come to equilibrium at 150◦C. When equilibrium is established, the pressure of IBr is 0.200 atm. What is the pressure of I2 at equilibrium?

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 19:10, aamu15
Which statement correctly describes the phosphate ion, ? it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge on the phosphorus atom. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge on the phosphorus atom.
Answers: 3

Chemistry, 23.06.2019 02:00, matthewsorrow02
What is the mass of 0.750 mole of aluminum oxide, al2o3?
Answers: 1

Chemistry, 23.06.2019 05:50, starfox5454
Which of the following isotopes has the same number of neutrons as phosphorus-31?
Answers: 1
Do you know the correct answer?
The equilibrium constant Kp for the reaction I2(g) + Br2(g) ⇀↽ 2 IBr(g) + 11.7 kJ is 280 at 150◦C. S...
Questions in other subjects:

Mathematics, 15.10.2019 22:40



Mathematics, 15.10.2019 22:40

History, 15.10.2019 22:40


History, 15.10.2019 22:40


Chemistry, 15.10.2019 22:40

Mathematics, 15.10.2019 22:40


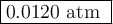
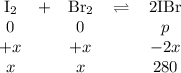
 }
}