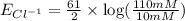Chemistry, 20.03.2020 01:57, Randomkid0973
1. Using the Nernst equation, calculate the equilibrium potential for Ca21 and for Cl2 from the following sets of data: a. Given [Ca21]o 5 1 mM, [Ca21]i 5 100 nM, find ECa21 b. Given [Cl2]o 5 110 mM, [Cl2]i 5 10 mM, find ECl2

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 11:00, snowprincess99447
Which statement is true about hcl? (5 points) select one: a. it is a salt because it increases the concentration of metallic ions. b. it is a salt because it is formed by the reaction of an acid and a base. c. it is an acid because it increases the concentration of hydroxyl ions. d. it is an acid because it increases the concentration of hydronium ions.
Answers: 1

Chemistry, 22.06.2019 12:10, yootmytoot
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution. calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1

Chemistry, 22.06.2019 22:20, icantspeakengles
Asuspension of yeast cells is being grown under anaerobic conditions such that glucose is degraded to ethanol and carbon dioxide. if one wishes to follow this process by monitoring the release of 14co2, at which positions in the glucose molecule would the 14c label need to be incorporated?
Answers: 2

Chemistry, 23.06.2019 05:00, jayden6467
How many moles are in 7.2 x 10^23 carbon molecules?
Answers: 1
Do you know the correct answer?
1. Using the Nernst equation, calculate the equilibrium potential for Ca21 and for Cl2 from the foll...
Questions in other subjects:


History, 11.01.2021 18:50

Computers and Technology, 11.01.2021 18:50


Mathematics, 11.01.2021 18:50



Business, 11.01.2021 18:50

Mathematics, 11.01.2021 18:50


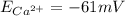 and
and 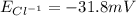
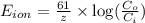
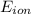 = equilibrium potential
= equilibrium potential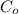 = concentration of the ion outside the cell
= concentration of the ion outside the cell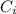 = concentration of the ion inside the cell
= concentration of the ion inside the cell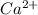 = 2
= 2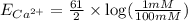
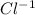 = 1
= 1