Chemistry, 20.03.2020 00:03, chickennbutt0730
In a different experiment, a student had 10.0 mL of 0.020 M HCl. Again, much to the surprise of the student, it took only 5.00 mL of 0.020 M strong base to react completely with the HCl. Explain why it took only half as much strong base to react with all of the HCl.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:00, hdjsjfjruejchhehd
Define dew point. i am writing this part to be able to ask the question
Answers: 1

Chemistry, 22.06.2019 13:00, monkeyrose1999
The molality of calcium chloride (cacl2) in an aqueous solution is 2.46 m. what is mole fraction of the solute?
Answers: 3

Chemistry, 22.06.2019 18:00, jalenclarke25
What volume would 2.25 moles of ne has occupy at stp?
Answers: 1

Do you know the correct answer?
In a different experiment, a student had 10.0 mL of 0.020 M HCl. Again, much to the surprise of the...
Questions in other subjects:


Mathematics, 06.11.2019 22:31


Mathematics, 06.11.2019 22:31






Social Studies, 06.11.2019 22:31


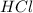 and the base and we have half of the volume therefore we have to keep in mind that we have a 1:2 molar ratio.
and the base and we have half of the volume therefore we have to keep in mind that we have a 1:2 molar ratio.