
Chemistry, 19.03.2020 23:58, johnnydenali67
A 2.45-g sample of strontium completely reacts with oxygen to form 2.89 g of strontium oxide. Use this data to calculate the mass percent composition of strontium in strontium oxide.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:50, nnaomii
What is the composition, in atom percent, of an alloy that consists of 4.5 wt% pb and 95.5 wt% sn? the atomic weights for pb and sn are 207.19 g/mol and 118.71 g/mol, respectively.(a) 2.6 at% pb and 97.4 at% sn(b) 7.6 at% pb and 92.4 at% sn(c)97.4 at% pb and 2.6 at% sn(d) 92.4 at% pb and 7.6 at% sn
Answers: 2

Chemistry, 21.06.2019 22:30, 20alondra04
In order to calculate the amount of heat transferred you must know the __ and specific heat of the material, as well as the change in temperature. a. volume b. density c. mass d. enthalpy
Answers: 1

Chemistry, 22.06.2019 00:10, goodygoodgirlygirl
Which of the following best describes the formation of plasma?
Answers: 1
Do you know the correct answer?
A 2.45-g sample of strontium completely reacts with oxygen to form 2.89 g of strontium oxide. Use th...
Questions in other subjects:


Mathematics, 18.08.2020 20:01

Mathematics, 18.08.2020 20:01

Mathematics, 18.08.2020 20:01

History, 18.08.2020 20:01






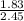 × 100
× 100 



