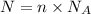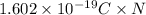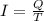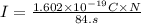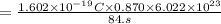Chemistry, 20.03.2020 00:03, F00Dislife
Suppose 870.mmol of electrons must be transported from one side of an electrochemical cell to another in 84. seconds. Calculate the size of electric current that must flow.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:10, shafferakr6
There are 6.022 x 10^23 atoms of hg in 1 mole of hg. the number of atoms in 4.5 moles of hg can be found by multiplying 4.5 by 6.022 x 10^23 a. 2.7 x 10^24 b. 27 x 10^23 c. 2.71 x10^24 d. 27.099 x 10^23
Answers: 3

Chemistry, 21.06.2019 20:50, deanlmartin
Choose all that apply. when creating a graph, you should: determine the x- and y- variables label the scale on the x- and y- axes plot the data points draw a line of best fit to represent the data trend
Answers: 1


Chemistry, 22.06.2019 09:20, kevinhernandez582
What will most likely happen when two bromine atoms bond together?
Answers: 3
Do you know the correct answer?
Suppose 870.mmol of electrons must be transported from one side of an electrochemical cell to anothe...
Questions in other subjects:

Mathematics, 24.07.2019 17:00

Biology, 24.07.2019 17:00

Mathematics, 24.07.2019 17:00

Mathematics, 24.07.2019 17:00


Biology, 24.07.2019 17:00




Computers and Technology, 24.07.2019 17:00