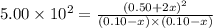T 745 K, the reaction below has an equilibrium constant (Kc) of 5.00 × 102. H2 (g) + I2 (g) ⇌ 2 HI (g) If a mixture of 0.10 mol H2, 0.10 mol I2 and 0.50 mol HI is sealed in a 1.00 L flask and heated to 745 K, what is the concentration of HI(g) when equilibrium is established?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:00, reeceslife481
What stress will shift the following equilibrium system to the left? n2(g) + 3h2(g) ⇌ 2nh3(g) adding more n2(g) adding more nh3(g) increasing the pressure of the system reducing the volume of the container
Answers: 1

Chemistry, 22.06.2019 18:00, ameliaxbowen7
Heat is the total potential energy of a substance that can be transferred. true false
Answers: 1

Chemistry, 22.06.2019 21:50, SoccerAllStar2
Liquid from a brewery fermentation contains 10% ethanol and 90% water. part of the fermentation product (50,000 kg/h) is pumped to a distillation column on the factory site. under current operating conditions, a distillate of 45% ethanol and 55% water is produced from the top of the column at a rate of one-tenth that of the feed. what is the composition of the waste "bottoms" from the still?
Answers: 2

Chemistry, 23.06.2019 00:30, hdhshshs741
An unknown insoluble substance displaced the water shown. it's mass is indicated on the triple beam balance. mass = a. 694 b. 693.5 c. 693.0 d.693.8
Answers: 1
Do you know the correct answer?
T 745 K, the reaction below has an equilibrium constant (Kc) of 5.00 × 102. H2 (g) + I2 (g) ⇌ 2 HI (...
Questions in other subjects:


Mathematics, 18.11.2019 09:31



Mathematics, 18.11.2019 09:31



English, 18.11.2019 09:31

Geography, 18.11.2019 09:31

History, 18.11.2019 09:31


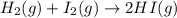
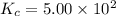
![K_c=\frac{[HI]^2}{[H_2][I_2]}](/tpl/images/0554/6132/62646.png)