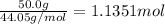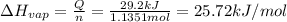29.2 kJ of heat is required to convert an 50.0 g sample of
acetaldehyde from the liquid to gas...

Chemistry, 19.03.2020 20:37, rexerlkman4145
29.2 kJ of heat is required to convert an 50.0 g sample of
acetaldehyde from the liquid to gas phase. The molecular
weight of acetaldehyde is 44.05 g/mol. What is the heat of
vaporization of acetaldehyde in kJ/mol?
kJ/mol

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, Queenquestion5967
When the following equation is balanced using the smallest possible integers, what is the coefficent of oxygen gas? c7h16o(g) + o2(g) → co2(g) + h2o(g) -1 -5 -8 -16 -21
Answers: 3

Chemistry, 22.06.2019 06:30, irvinbhangal2
What effect might melting sea ice have for people who live in coastal areas?
Answers: 1

Chemistry, 22.06.2019 14:30, Dreynolds1667
100 grams of molten lead (600°c) is used to make musket balls. if the lead shot is allowed to cool to room temperature (21°c), what is the change in entropy (in j/k) of the lead? (for the specific heat of molten and solid lead use 1.29 j/g⋅°c; the latent heat of fusion and the melting point of lead are 2.45 × 104 j/kg and 327°c, respectively.)
Answers: 1

Do you know the correct answer?
Questions in other subjects:


Mathematics, 12.01.2020 12:31



Mathematics, 12.01.2020 12:31




History, 12.01.2020 12:31