
Chemistry, 19.03.2020 08:29, RealSavage4Life
P is the pressure in atmospheres (atm), V is the volume in liters (L), n is the number of moles, R is the gas constant (0.0821 L∙atm/(mol∙K)), and T is the temperature in Kelvins (K). Consider the following conditions: a sample of neon gas was under 3.0 atm of pressure, a volume of 570 mL with a temperature of 75 °C. Assume you are going to use the ideal gas law to solve for the unknown variable. What variable are you solving for? Are all of variables in the correct units? If not, which variable needs to be converted to the correct units?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:00, pollywallythecat
Your friend offers to show you an intrusive igneous rock. which of the following would you expect to see?
Answers: 1


Chemistry, 22.06.2019 13:20, alejandra340
Can someone me with 3 and 4 plz. this is for masteries test.
Answers: 2
Do you know the correct answer?
P is the pressure in atmospheres (atm), V is the volume in liters (L), n is the number of moles, R i...
Questions in other subjects:

History, 17.12.2020 01:00




Mathematics, 17.12.2020 01:00


Mathematics, 17.12.2020 01:00

History, 17.12.2020 01:00


Mathematics, 17.12.2020 01:00


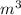
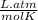
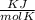
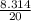 = 0.41
= 0.41 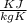
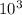 × 0.00057 = m × 0.41 × 348
× 0.00057 = m × 0.41 × 348 



