
A piece of iron (mass = 25.0 g) at 398 K is placed in a styrofoam coffee cup containing 25.0 mL of water at 298 K. Assuming that no heat is lost to the cup or the surroundings, what will the final temperature of the water be? The specific heat capacity of iron = 0.449 J/g°C and water = 4.18 J/g°C.

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 02:30, BornAdopted21
Which piece of equipment would me most useful for measuring the volume of some water? a. pan balance b. graduated cylinder c. tweezers d. flask quick
Answers: 2

Chemistry, 22.06.2019 05:50, mrylenastewart
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3 -> a2cr(no3)3 + a3ag2so4
Answers: 1

Do you know the correct answer?
A piece of iron (mass = 25.0 g) at 398 K is placed in a styrofoam coffee cup containing 25.0 mL of w...
Questions in other subjects:


Mathematics, 13.11.2019 22:31


Social Studies, 13.11.2019 22:31


Mathematics, 13.11.2019 22:31

History, 13.11.2019 22:31




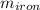 = 25 gm
= 25 gm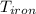 = 398 K
= 398 K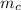 = 25 ml = 25 gm
= 25 ml = 25 gm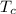 = 298 K
= 298 K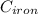 = 0.449
= 0.449 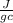
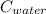 = 4.18
= 4.18 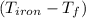 =
= 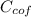 ×
× 
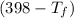 = 25 × 4.18 ×
= 25 × 4.18 × 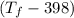
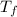 = 9.3 (
= 9.3 ( 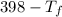 = 9.3
= 9.3 



