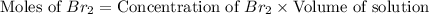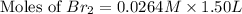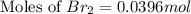Chemistry, 19.03.2020 01:16, blackwhiteroses383
Consider the reaction. 2 HBr(g) ¡ H2(g) + Br2(g) a. Express the rate of the reaction in terms of the change in concentration of each of the reactants and products. b. In the first 25.0 s of this reaction, the concentration of HBr drops from 0.600 M to 0.512 M. Calculate the average rate of the reaction during this time interval. c. If the volume of the reaction vessel in part b is 1.50 L, what amount of Br2 (in moles) forms during the first 15.0 s of the reaction?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 21:00, taylorlanehart
Use the measurements in the table to determine which unidentified metal has the highest density. metal volume mass a 10.5 cm3 122 g b 14.2 cm3 132 g c 16.1 cm3 115 g d 12.7 cm3 126 g
Answers: 2

Chemistry, 22.06.2019 21:20, carlydays4403
The organs inside the body and how they function together
Answers: 3


Chemistry, 23.06.2019 13:30, sophie5988
Where are electrons with the lowest energy found? in the nucleus farthest from the nucleus outside the atom closest to the nucleus
Answers: 1
Do you know the correct answer?
Consider the reaction. 2 HBr(g) ¡ H2(g) + Br2(g) a. Express the rate of the reaction in terms of the...
Questions in other subjects:



Mathematics, 22.06.2020 21:57

Advanced Placement (AP), 22.06.2020 21:57

English, 22.06.2020 21:57






![Rate=-\frac{1}{2}\frac{d[HBr]}{dt}=+\frac{d[H_2]}{dt}=+\frac{d[Br_2]}{dt}](/tpl/images/0553/1566/27c4e.png)
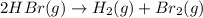
![\text{Rate of disappearance of }HBr=-\frac{1}{2}\frac{d[HBr]}{dt}](/tpl/images/0553/1566/d63dd.png)
![\text{Rate of disappearance of }H_2=+\frac{d[H_2]}{dt}](/tpl/images/0553/1566/eb73c.png)
![\text{Rate of formation of }Br_2=+\frac{d[Br_2]}{dt}](/tpl/images/0553/1566/30b3c.png)
![\text{Average rate}=-\frac{1}{2}\frac{d[HBr]}{dt}](/tpl/images/0553/1566/79555.png)
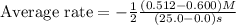
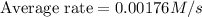
![\frac{d[Br_2]}{dt}=0.00176M/s](/tpl/images/0553/1566/78ef0.png)
![\frac{d[Br_2]}{15.0s}=0.00176M/s](/tpl/images/0553/1566/22daf.png)
![[Br_2]=0.00176M/s\times 15.0s](/tpl/images/0553/1566/5d9c4.png)
![[Br_2]=0.0264M](/tpl/images/0553/1566/42226.png)