
Chemistry, 18.03.2020 13:30, contrerasdaisy100
How many moles of HNO3 are present if 4.90×10−2 mol of Ba(OH)2 was needed to neutralize the acid solution?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, brittanygibson2812
Aballoon inflated with three breaths of air has a volume of 1.7 l. at the same temperature and pressure, what is the volume of the balloon if five more same-sized breaths are added to the balloon?
Answers: 3


Chemistry, 22.06.2019 21:30, imalexiscv
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1
Do you know the correct answer?
How many moles of HNO3 are present if 4.90×10−2 mol of Ba(OH)2 was needed to neutralize the acid sol...
Questions in other subjects:


Mathematics, 06.01.2021 03:20

Mathematics, 06.01.2021 03:20

Health, 06.01.2021 03:20


Computers and Technology, 06.01.2021 03:20

Mathematics, 06.01.2021 03:20




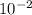 mol of Ba(OH)₂ was needed to neutralize the acid solution
mol of Ba(OH)₂ was needed to neutralize the acid solution




