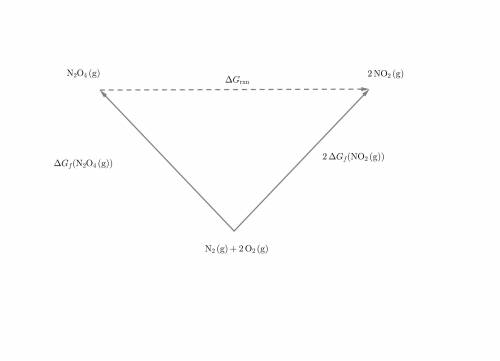
Chemistry, 18.03.2020 00:50, loganparrish2488
Select the correct answer,
What is the AG for the following reaction at 25°C?
N204(8) 2N02(9)
Given:
N20409): AG= 98.28 kJ/mol
NO2(9): AG= 51.8 kJ/mol
OA 5.32 kJ/mol
OB. 10.64 kJ/mol
OC -5.32 kj/mol
OD. -10.64 kJ/mol

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 19:20, halledoll2002
Anyone who's in connections academy chemistry b have the factors that affect the rate of a reaction portfolio already done?
Answers: 3

Chemistry, 22.06.2019 20:00, aksambo4707
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1

Chemistry, 23.06.2019 00:10, Rubendelarosa1529
Covalent compounds: mastery test select the correct answer what is formed when atoms join together with a covalent bond? a. an ion b. a molecule c. a neutral atom d. a noble gas
Answers: 3
Do you know the correct answer?
Select the correct answer,
What is the AG for the following reaction at 25°C?
N204(8) 2N...
What is the AG for the following reaction at 25°C?
N204(8) 2N...
Questions in other subjects:



Biology, 18.01.2020 11:31

Mathematics, 18.01.2020 11:31


Biology, 18.01.2020 11:31



Mathematics, 18.01.2020 11:31

Mathematics, 18.01.2020 11:31


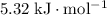 .
. 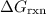 can be found from the Gibbs Energy of formation
can be found from the Gibbs Energy of formation 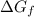 :
: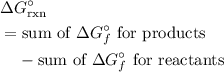 .
.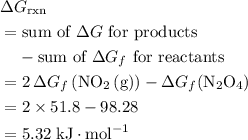 .
.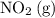 molecules on the product side of this reaction, the
molecules on the product side of this reaction, the 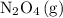 to
to 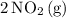 . To find that value, invert
. To find that value, invert 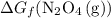 (the arrow on the left-hand side) and add that to
(the arrow on the left-hand side) and add that to 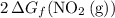 (the arrow on the right-hand side.) The resultant value would be the dashed arrow, which stands for
(the arrow on the right-hand side.) The resultant value would be the dashed arrow, which stands for 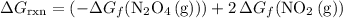 .
.