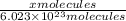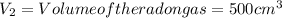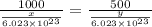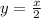Chemistry, 17.03.2020 22:04, sidneydominguez2323
1000 cm3 of hydrogen gas (hydrogen molecules, H2) contains x number of molecules at room temperature and pressure. Determine the number of atoms in 500 cm3 of radon gas (radon atoms) at the same temperature and pressure

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:50, giiffnlojd
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3


Chemistry, 22.06.2019 10:40, yfgkeyonna
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2

Chemistry, 22.06.2019 18:50, christhegreat1
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1
Do you know the correct answer?
1000 cm3 of hydrogen gas (hydrogen molecules, H2) contains x number of molecules at room temperature...
Questions in other subjects:



Mathematics, 08.12.2020 21:00

Mathematics, 08.12.2020 21:00

Arts, 08.12.2020 21:00


Mathematics, 08.12.2020 21:00

Mathematics, 08.12.2020 21:00


Social Studies, 08.12.2020 21:00


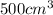 of radon gas (radon atoms) at the same temperature and pressure is
of radon gas (radon atoms) at the same temperature and pressure is 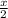
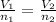
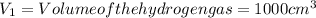
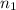 = moles of hydrogen gas =
= moles of hydrogen gas =