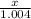Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:10, gabriellestaleyga16
How many moles of gas are present if p=11 atm, v=12l, t=185k?
Answers: 1

Chemistry, 22.06.2019 12:00, daytonalive83481
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Do you know the correct answer?
Calculate the mass of hydrogen formed when 27 g of aluminum reacts with excess hydrochloric acid acc...
Questions in other subjects:

Mathematics, 25.04.2021 19:30


Mathematics, 25.04.2021 19:30

English, 25.04.2021 19:30

English, 25.04.2021 19:30



Arts, 25.04.2021 19:30

Mathematics, 25.04.2021 19:30


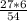 = 3 g of H₂.
= 3 g of H₂.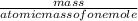
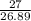
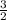 =
=