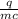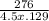Chemistry, 17.03.2020 20:02, fansofboys
A 4.50 g nugget of pure gold absorbed 276 J of heat. the initial temperature was 25.0 degrees C. what was the final temperature?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:30, ruleolivas
Asample of ammonia reacts with oxygen as shown. 4nh3(g) + 5o2(g) 4no(g) + 6h2o(g) what is the limiting reactant if 4.0 g of nh3 react with 8.0 g of oxygen? o2 because it produces only 0.20 mol of no. nh3 because it produces only 0.20 mol of no. o2 because it produces two times less no than nh3. nh3 because it produces three times more no than o2.
Answers: 3


Chemistry, 22.06.2019 10:30, jahmira96
Use this information to determine the number of calends electrons in the atoms. which of the following correctly compares the stability of the two atoms? a) both are unreactive b) both are highly reactive c) a is unreactive and d is reactive d) a is reactive and d is unreactive
Answers: 2
Do you know the correct answer?
A 4.50 g nugget of pure gold absorbed 276 J of heat. the initial temperature was 25.0 degrees C. wha...
Questions in other subjects:

Chemistry, 05.09.2020 19:01



History, 05.09.2020 19:01



Mathematics, 05.09.2020 19:01



Chemistry, 05.09.2020 19:01