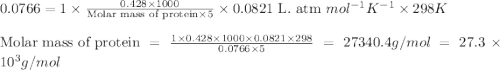Chemistry, 17.03.2020 05:41, meramera50
428. mg of an unknown protein are dissolved in enough solvent to make of solution. The osmotic pressure of this solution is measured to be at . Calculate the molar mass of the protein. Round your answer to significant digits.

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 23.06.2019 02:00, hermesrobles
Which would freeze at a higher temperature: the great salt lake or lake tahoe? a. lake tahoe would freeze at a higher temperature. b. the great salt lake would freeze at a higher temperature. c. both lakes would freeze at the same temperature.
Answers: 2

Chemistry, 23.06.2019 04:20, lelliott86
The lewis diagrams for magnesium and fluorine are shown below. what is the correct chemical formula for magnesium fluoride? a. mgf b. mg2f c. mgf2 d. mg2f3
Answers: 1
Do you know the correct answer?
428. mg of an unknown protein are dissolved in enough solvent to make of solution. The osmotic press...
Questions in other subjects:


Physics, 08.03.2020 19:48

Mathematics, 08.03.2020 19:49






Mathematics, 08.03.2020 19:52


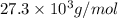
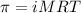
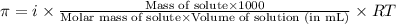
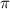 = osmotic pressure of the solution = 0.0766 atm
= osmotic pressure of the solution = 0.0766 atm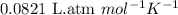
![25^oC=[273+25]=298K](/tpl/images/0550/3624/6a9f9.png)