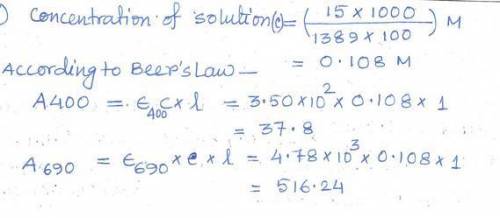In order to wash out the impurity, you looked up some sketchy method from 1952 which Hank Pym wrote during his Ph. D. under E. J. Corey. The method requires you add HCl to the mixture. After you rid your sample of any impurities, you take some pH paper and find out your solution has a pH of 3.0. Assuming the pKa of Pym Particles is 4.5 and that the molar absorptivity of the protonated Pym Particle is 4.82 * 102 L/mol*cm and 8.54 * 103 L/mol*cm for 400 nm and 690 nm respectively, calculate the expected A400 and A690 of the solution.

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 04:30, terrancebest
Which is a chemical property of iron? a. it forms iron oxide (rust) when exposed to moisture and air. b. it is a gray–black metal that is hard to the touch. c. it has a melting point of 2795°f (1536°c). d. it is a good conductor of heat
Answers: 2

Chemistry, 22.06.2019 07:30, 10040813
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 12:10, purplefish53
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3
Do you know the correct answer?
In order to wash out the impurity, you looked up some sketchy method from 1952 which Hank Pym wrote...
Questions in other subjects:

Mathematics, 29.12.2019 23:31

Mathematics, 29.12.2019 23:31

Business, 29.12.2019 23:31


Mathematics, 29.12.2019 23:31


Mathematics, 29.12.2019 23:31

Computers and Technology, 29.12.2019 23:31

Business, 29.12.2019 23:31

Mathematics, 29.12.2019 23:31