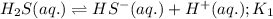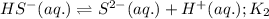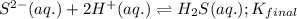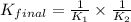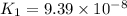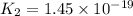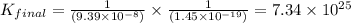Chemistry, 17.03.2020 04:33, channarlawassociate
Given the two reactions H2S(aq)⇌HS−(aq)+H+(aq), H2S(aq)⇌HS−(aq)+H+(aq), K1K1K_1 = 9.39×10−8, and HS−(aq)⇌S2−(aq)+H+(aq), HS−(aq)⇌S2−(aq)+H+(aq), K2K2K_2 = 1.45×10−19, what is the equilibrium constant KfinalKfinalK_final for the following reaction? S2−(aq)+2H+(aq)⇌H2S(aq)

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, giraffegurl
If 34.2 grams of lithium react with excess water, how many liters of hydrogen gas can be produced at 299 kelvin and 1.21 atmospheres? 2 li (s) + 2 h2o (l) yields 2 lioh (aq) + h2 (g)
Answers: 3


Do you know the correct answer?
Given the two reactions H2S(aq)⇌HS−(aq)+H+(aq), H2S(aq)⇌HS−(aq)+H+(aq), K1K1K_1 = 9.39×10−8, and HS−...
Questions in other subjects:

Mathematics, 19.11.2020 05:50

History, 19.11.2020 05:50




Mathematics, 19.11.2020 05:50





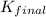 for the net reaction is
for the net reaction is