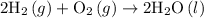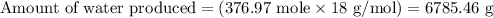Chemistry, 17.03.2020 03:30, shiyaharper
For the reactants represented by the equation 2H2 + O2 > 2H2O, how many grams of water are produced from 376.97 mol of hydrogen?

Answers: 1
Other questions on the subject: Chemistry




Chemistry, 22.06.2019 18:00, rodriguezscarlet1713
Hydrogenation reactions, in which h2 and an "unsaturated" organic compound combine, are used in the food, fuel, and polymer industries. in the simplest case, ethene (c2h4) and h2 form ethane (c2h6). if 140 kj is given off per mole of c2h4 reacting, how much heat (in mj) is released when 12 kg of c2h6 forms?
Answers: 2
Do you know the correct answer?
For the reactants represented by the equation 2H2 + O2 > 2H2O, how many grams of water are produc...
Questions in other subjects:



Arts, 10.05.2021 18:00



History, 10.05.2021 18:00



Mathematics, 10.05.2021 18:00

Mathematics, 10.05.2021 18:00