
Chemistry, 16.03.2020 23:45, alejandramirand9836
A cylinder with a moveable piston contains 92g of Nitrogen. The external pressure is constant at 1.00 atm. The initial temperature is 200K. When the temperature is decreased by 85 K, by putting it in a lower temperature freezer, the volume will decrease, according to the Ideal Gas Law. Calculate the work for this process. Express your answer in J. The conversion factor between liter atmospheres and joules is 101.3 J

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:00, smhrosepetals
Which solution is an example of a nonelectrolyte? a. iodine in hexane b. sodium nitrate in waterc. acetic acid in waterd. hydrogen chloride in water
Answers: 2

Chemistry, 22.06.2019 01:00, jescanarias22
Which statement correctly describes potassium iodide, ki? there is a one-to-one ratio of potassium ions to iodide ions. potassium gains electrons and iodine loses electrons during the reaction. the lattice is held together by potassium anions and iodide cations.
Answers: 1

Do you know the correct answer?
A cylinder with a moveable piston contains 92g of Nitrogen. The external pressure is constant at 1.0...
Questions in other subjects:

Mathematics, 24.02.2021 21:50

Mathematics, 24.02.2021 21:50


Mathematics, 24.02.2021 21:50

Spanish, 24.02.2021 21:50

History, 24.02.2021 21:50

Mathematics, 24.02.2021 21:50

Mathematics, 24.02.2021 21:50

History, 24.02.2021 21:50


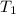 = 200 K
= 200 K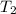 = 200 - 85 = 115 K
= 200 - 85 = 115 K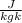
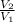 =
= 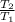 ------------ ( 1 )
------------ ( 1 )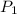
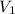 = m R
= m R 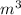
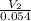 =
= 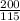
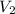 = 0.094
= 0.094 



