
Chemistry, 16.03.2020 22:34, McGregorshamara
Write the dissolution reaction for iron(III) nitrate in water. Use the pull-down menus to specify the state of each reactant and product. + Is iron(III) nitrate considered soluble or not soluble ? ... ... A. Soluble ... B. Not soluble Based upon this, the equilibrium constant for this reaction will be: ... ... A. Greater than 1 ... B. Less than 1

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, arodavoarodavo
The table lists pressure and volume values for a particular gas. which is the best estimate for the value of v at p = 7.0 × 103 pascals?
Answers: 3

Chemistry, 22.06.2019 08:00, hdjsjfjruejchhehd
Define dew point. i am writing this part to be able to ask the question
Answers: 1

Chemistry, 22.06.2019 09:00, alydiale584
Particles vibrate in a rigid structure and do not move relative to their neighbors.
Answers: 1

Do you know the correct answer?
Write the dissolution reaction for iron(III) nitrate in water. Use the pull-down menus to specify th...
Questions in other subjects:


Mathematics, 15.01.2021 02:50

Mathematics, 15.01.2021 02:50

Arts, 15.01.2021 02:50




Mathematics, 15.01.2021 02:50


Mathematics, 15.01.2021 02:50


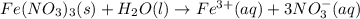
![K_{eq} = [Fe^{3+}][(NO_{3})_{3}]^{3}](/tpl/images/0549/4240/1e6b7.png)




