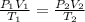A child has a toy balloon with a volume of 1.80 L. The temperature of the balloon when it was filled was 293 K at a pressure of 101.3 kPa. If the child were to let go of the balloon and it rose 3 kilometers into the sky where the pressure of 67.6 kPa and the temperature is 263 K, what would the new volume of the balloon be?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:00, penelopymorales24
Match each object to its description: a. coma of a comet b. comet tail c. oort cloud haze surrounding a nucleus created by solar wind. hypothetical sphere around the solar system
Answers: 1


Chemistry, 22.06.2019 09:00, mercymain1014
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1

Chemistry, 22.06.2019 14:00, emilyproce
In the space, show a correct numerical setup for calculating the number of moles of co2 present in 11 grams of co2
Answers: 1
Do you know the correct answer?
A child has a toy balloon with a volume of 1.80 L. The temperature of the balloon when it was filled...
Questions in other subjects:

English, 30.10.2021 01:00


Mathematics, 30.10.2021 01:00



SAT, 30.10.2021 01:00


Mathematics, 30.10.2021 01:00


Computers and Technology, 30.10.2021 01:00