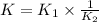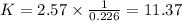Chemistry, 16.03.2020 20:53, cravetyler3989
Wo reactions and their equilibrium constants are given. A + 2 B − ⇀ ↽ − 2 C K 1 = 2.57 2 C − ⇀ ↽ − D K 2 = 0.226 A+2B↽−−⇀2CK1=2.572C↽−−⇀DK2=0.226 Calculate the value of the equilibrium constant for the reaction D − ⇀ ↽ − A + 2 B .

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:00, alydiale584
Particles vibrate in a rigid structure and do not move relative to their neighbors.
Answers: 1

Chemistry, 22.06.2019 10:00, melissa9882
A50.0g sample of liquid water at 0.0 c ends up as ice at -20.0 c. how much energy is involved in this change?
Answers: 1

Chemistry, 22.06.2019 11:00, daniel1480
Problem page combustion of hydrocarbons such as pentane ( c5 h12 ) produces carbon dioxide, a "greenhouse gas." greenhouse gases in the earth's atmosphere can trap the sun's heat, raising the average temperature of the earth. for this reason there has been a great deal of international discussion about whether to regulate the production of carbon dioxide.(a) write a balanced chemical equation, including physical state symbols, for the combustion of liquid pentane into gaseous carbon dioxide and gaseous water. (b) suppose 0.350 kg of pentane are burned in air at a pressure of exactly 1 atm and a temperature of 20.0 degree c. calculate the volume of carbon dioxide gas that is produced. be sure your answer has the correct number of significant digits.
Answers: 2
Do you know the correct answer?
Wo reactions and their equilibrium constants are given. A + 2 B − ⇀ ↽ − 2 C K 1 = 2.57 2 C − ⇀ ↽ − D...
Questions in other subjects:



History, 18.01.2020 22:31

History, 18.01.2020 22:31

History, 18.01.2020 22:31

Spanish, 18.01.2020 22:31




Mathematics, 18.01.2020 22:31


![A+2B\xrightarrow[]{K_1} 2C](/tpl/images/0549/1251/bf2d5.png)
![2C\xrightarrow[]{K_2} D](/tpl/images/0549/1251/8564b.png)
![D\xrightarrow[]{K} A+2B](/tpl/images/0549/1251/d3cc9.png)