
Chemistry, 16.03.2020 20:27, ronniethefun
In a coffee-cup calorimeter experiment, 10.00 g of a soluble ionic compound was added to the calorimeter containing 75.0 g H2O initially at 23.2°C. The temperature of the water increased to 31.8°C. What was the change in enthalpy for the dissolution of the compound? Give your answer in units of joules per gram of compound. Assume that the specific heat of the solution is the same as that of pure water, 4.18 J ⁄ (g ⋅ °C).

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:30, junkmailemail42
Which element is the least metallic between cadmium, silver, zinc, or iron?
Answers: 1

Chemistry, 22.06.2019 14:30, belindajolete
How do temperature and salinity affect deepwater currents? as temperatures and salinity levels of water increase, the water rises to the surface where it creates currents as it moves to colder regions. they create changes in wind direction, moving denser water in the same direction as the wind and causing the deepwater circulation patterns found in the ocean. they equalize the forces on undersea currents caused by the coriolis effect as they replace more dense water with less dense water. they create density differences that cause dense deepwater currents to flow toward the equator where they displace less dense, warmer water above them.
Answers: 2


Chemistry, 22.06.2019 22:30, itsmaddierae11
Which of the following molecules is polar? c3h7oh c2h5cooh
Answers: 1
Do you know the correct answer?
In a coffee-cup calorimeter experiment, 10.00 g of a soluble ionic compound was added to the calorim...
Questions in other subjects:

Mathematics, 11.10.2019 08:00

English, 11.10.2019 08:00

Mathematics, 11.10.2019 08:00

Mathematics, 11.10.2019 08:00


English, 11.10.2019 08:00

English, 11.10.2019 08:00

Biology, 11.10.2019 08:00


Mathematics, 11.10.2019 08:00


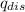 ) is equal to the heat of water (
) is equal to the heat of water (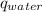 ).
). 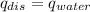 = ΔH/m= 2696.1 J/10.00 g= 269.61 J/g
= ΔH/m= 2696.1 J/10.00 g= 269.61 J/g



