
Chemistry, 16.03.2020 20:09, ginareyes0423
Calculate ΔHrxnΔHrxn for the following reaction: 3C(s)+4H2(g)→C3H8(l)3C(s)+4H2(g)→C3 H8(l) Use the following reactions and given ΔHΔH values: C3H8(l)+5O2(g)→3CO2(g)+4H2O(g),ΔHC( s)+O2(g)→CO2(g),ΔH2H2(g)+O2(g)→2H2O (g),ΔH===−2026.6kJ−393.5kJ−483.5kJ

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:20, Brooke7644
Calcium hydride (cah2) reacts with water to form hydrogen gas: cah2(s) + 2h2o(l) → ca(oh)2(aq) + 2h2(g) how many grams of cah2 are needed to generate 45.0 l of h2 gas at a pressure of 0.995 atm and a temperature of 32 °c?
Answers: 2

Chemistry, 22.06.2019 01:00, bettybales1986
According to the tide table below what time of day will the highest tide occur?
Answers: 1

Chemistry, 22.06.2019 15:00, MilanPatel
How is the shape of the poem “peer” connected to its meaning?
Answers: 2
Do you know the correct answer?
Calculate ΔHrxnΔHrxn for the following reaction: 3C(s)+4H2(g)→C3H8(l)3C(s)+4H2(g)→C3 H8(l) Use the f...
Questions in other subjects:



Mathematics, 02.09.2019 00:30



Mathematics, 02.09.2019 00:30


English, 02.09.2019 00:30

Chemistry, 02.09.2019 00:30

Biology, 02.09.2019 00:30


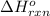 for the reaction is -120.9 kJ.
for the reaction is -120.9 kJ.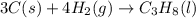
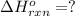
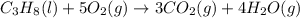
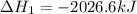
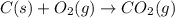
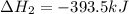 ( × 3)
( × 3)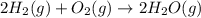
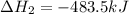 ( × 2)
( × 2)![\Delta H^o_{rxn}=[1\times (-\Delta H_1)]+[3\times \Delta H_2]+[2\times \Delta H_3]](/tpl/images/0548/9833/b4dbe.png)
![\Delta H^o_{rxn}=[(1\times -(-2026.6))+(3\times (-393.5))+(2\times (-483.5))]=-120.9kJ](/tpl/images/0548/9833/4ea2a.png)




