

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, sandersmakaylaovq5vu
The balanced chemical equation for this lab is: 3cucl2(aq) + 2al(s) 3cu(s) + 2alcl3(aq) if 10.5 g copper chloride react with 12.4 g aluminum, what is the limiting reactant?
Answers: 3

Chemistry, 22.06.2019 05:50, mrylenastewart
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3 -> a2cr(no3)3 + a3ag2so4
Answers: 1

Chemistry, 22.06.2019 09:10, chloeholt123
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 10:10, babyphoraaaaa
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate, m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3
Do you know the correct answer?
The equilibrium constant, Kc, for the following reaction is 77.5 at 600 K. CO(g) Cl2(g) COCl2(g) Cal...
Questions in other subjects:


Biology, 12.04.2021 18:20


Biology, 12.04.2021 18:20

Mathematics, 12.04.2021 18:20

English, 12.04.2021 18:20





![[COCl_2]_{eq}=0.424M](/tpl/images/0548/6803/95daf.png)
![[Cl_2]_{eq}=0.074M](/tpl/images/0548/6803/a792c.png)
![[CO]_{eq}=0.074M](/tpl/images/0548/6803/01ccd.png)

![Keq=\frac{[COCl_2]}{[CO][Cl_2]}=77.5](/tpl/images/0548/6803/db058.png)
![[CO]_0=[Cl_2]_0=\frac{0.498mol}{1L}=0.498M](/tpl/images/0548/6803/f6c6e.png)
 is introduced due to the chemical reaction, considering the stoichiometry, the law of mass action turns out:
is introduced due to the chemical reaction, considering the stoichiometry, the law of mass action turns out: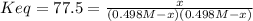

![[COCl_2]_{eq}=x=0.424M](/tpl/images/0548/6803/1f335.png)
![[Cl_2]_{eq}=0.498M-x=0.498M-0.424M=0.074M](/tpl/images/0548/6803/f144d.png)
![[CO]_{eq}=0.498M-x=0.498M-0.424M=0.074M](/tpl/images/0548/6803/bb482.png)




