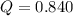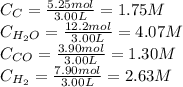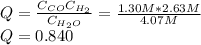The following reaction was carried out in a 3.00 L reaction vessel at 1100 K:
C(s)+H2O(...

Chemistry, 16.03.2020 16:41, lizzet2557
The following reaction was carried out in a 3.00 L reaction vessel at 1100 K:
C(s)+H2O(g) --> CO(g)+H2(g)
If during the course of the reaction, the vessel is found to contain 5.25 mol of C, 12.2 mol of H2O, 3.90 mol of CO, and 7.90 mol of H2, what is the reaction quotient Q?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:00, pettygirl13
Describe the particles of all three phases of matter in the root beer float. (how are the particles arranged and moving? )
Answers: 3


Chemistry, 22.06.2019 06:00, mbrisen7420
Compare and contrast physical changes with chemical changes.
Answers: 3

Chemistry, 23.06.2019 09:30, noeliaalvarado
Which element below could be and isotope of this atom
Answers: 1
Do you know the correct answer?
Questions in other subjects:

Mathematics, 02.03.2021 17:30

Social Studies, 02.03.2021 17:30

Mathematics, 02.03.2021 17:30

Mathematics, 02.03.2021 17:30






Advanced Placement (AP), 02.03.2021 17:30