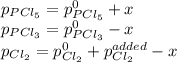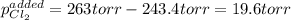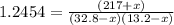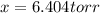Chemistry, 12.03.2020 21:01, jsmith4184
An equilibrium mixture of PCl 5 ( g ) , PCl 3 ( g ) , and Cl 2 ( g ) has partial pressures of 217.0 Torr, 13.2 Torr, and 13.2 Torr, respectively. A quantity of Cl 2 ( g ) is injected into the mixture, and the total pressure jumps to 263.0 Torr at the moment of mixing. The system then re-equilibrates. The chemical equation for this reaction is PCl 3 ( g ) + Cl 2 ( g ) − ⇀ ↽ − PCl 5 ( g ) Calculate the new partial pressures, P , after equilibrium is reestablished.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, bartfrank447
Joseph has hypothesized that sound travels in waves. if he were following the scientific method, what should he do next? a. ask a question. b. test the hypothesis. c. study the results. d. tell other scientists about his hypothesis.
Answers: 1

Chemistry, 22.06.2019 01:00, chrisxxxrv24
What are the variables in gay-lussac’s law? pressure and volume pressure, temperature, and volume pressure and temperature volume, temperature, and moles of gas
Answers: 1

Chemistry, 22.06.2019 09:30, jewelz5887
1. explain hydrogen peroxide, h 2 o 2 properties and decomposition reaction. 2. describe how each of the following natural cycles plays a part in earth’s climate system. (a) the water cycle (b) the carbon cycle
Answers: 1

Chemistry, 22.06.2019 12:00, angtrevv
In a laboratory, 1.55mg of an organic compound containing carbon, hydrogen, and oxygen is burned for analysis. this combustion resulted in the formation of 1.45mg of carbon dioxide and .89 mg of water. what is the empirical formula for this compound?
Answers: 1
Do you know the correct answer?
An equilibrium mixture of PCl 5 ( g ) , PCl 3 ( g ) , and Cl 2 ( g ) has partial pressures of 217.0...
Questions in other subjects:


History, 25.06.2019 06:30




Mathematics, 25.06.2019 06:30




Biology, 25.06.2019 06:30


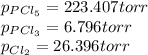
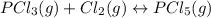
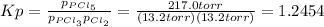
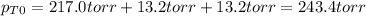
 owing to the chlorine's addition, turn out:
owing to the chlorine's addition, turn out: