
Chemistry, 12.03.2020 20:27, eduardoma2902
At a certain temperature the value of the equilibrium constant, Kc, is 1.27 for the reaction. 2As (s) + 3H2 (g) ⇌ 2AsH3 (g) What is the value of Kc for the following reaction? AsH3 (g) ⇌ As (s) + 3/2H2 (g)

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:40, yah2muchh
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 20:30, trevorhenyan51
What is a difference between a mixture of elements and a mixture of compounds
Answers: 1

Chemistry, 23.06.2019 00:30, danielmartinez024m
Maya wrote if you step to describe how carbon circulates between the atmosphere and living organisms
Answers: 1
Do you know the correct answer?
At a certain temperature the value of the equilibrium constant, Kc, is 1.27 for the reaction. 2As (s...
Questions in other subjects:

Mathematics, 02.09.2020 23:01


Mathematics, 02.09.2020 23:01



Mathematics, 02.09.2020 23:01

Physics, 02.09.2020 23:01




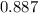
![Kc=\frac{[AsH_3]^2}{[As]^2[H_2] ^3}=1.27](/tpl/images/0545/3503/09ec8.png)
![Kc=\frac{[As][H_2] ^{3/2}}{[AsH_3]}](/tpl/images/0545/3503/b2ae8.png)
![\sqrt{Kc} =\sqrt{\frac{[AsH_3]^2}{[As]^2[H_2] ^3}} =\sqrt{1.27}](/tpl/images/0545/3503/dc32b.png)
![\frac{\sqrt{[AsH_3]^2} }{\sqrt{[As]^2} \sqrt{[H_2] ^3} } =\sqrt{1.27}](/tpl/images/0545/3503/19f81.png)
![\sqrt{1.27}=\frac{[AsH_3]}{[As][H_2] ^{3/2}}](/tpl/images/0545/3503/e4fe4.png)
![Kc'=\frac{1}{\sqrt{1.27} }=\frac{[As][H_2] ^{3/2}}{[AsH_3]}=0.887](/tpl/images/0545/3503/a88a2.png)





