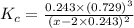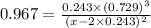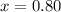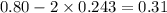The equilibrium constant, Kc, for the following reaction is 0.967 at 650 K. 2NH3(g) N2(g) 3H2(g) When a sufficiently large sample of NH3(g) is introduced into an evacuated vessel at 650 K, the equilibrium concentration of H2(g) is found to be 0.729 M. Calculate the concentration of NH3 in the equilibrium mixture. M

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:30, coreyslotte
Use examples from the article to explain one positive and one negative effect that chemistry has had on society.
Answers: 2

Chemistry, 22.06.2019 10:40, trinityanne1738
Asolid that forms and separates from a liquid mixture is called
Answers: 2

Chemistry, 22.06.2019 15:10, kolbehoneyman
The ozone molecule o3 has a permanent dipole moment of 1.8×10−30 cm. although the molecule is very slightly bent-which is why it has a dipole moment-it can be modeled as a uniform rod of length 2.5×10−10 m with the dipole moment perpendicular to the axis of the rod. suppose an ozone molecule is in a 8000 n/c uniform electric field. in equilibrium, the dipole moment is aligned with the electric field. but if the molecule is rotated by a small angle and released, it will oscillate back and forth in simple harmonic motion. what is the frequency f of oscillation?
Answers: 2

Chemistry, 22.06.2019 16:30, ddmoorehouseov75lc
Correct relationship between molecular formula and empirical formula
Answers: 1
Do you know the correct answer?
The equilibrium constant, Kc, for the following reaction is 0.967 at 650 K. 2NH3(g) N2(g) 3H2(g) Whe...
Questions in other subjects:

Mathematics, 07.10.2019 07:30


Chemistry, 07.10.2019 07:30

Health, 07.10.2019 07:30


Social Studies, 07.10.2019 07:30



Mathematics, 07.10.2019 07:30


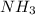 in the equilibrium mixture is 0.31 M
in the equilibrium mixture is 0.31 M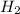 = 0.729 M
= 0.729 M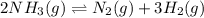
![K_c=\frac{[y]\times [3y]^3}{[x-2y]^2}](/tpl/images/0544/6710/3f7db.png)