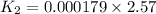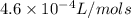Chemistry, 12.03.2020 03:55, GOOBER3838
The reaction between nitrogen dioxide and carbon monoxide is NO2(g)+CO(g)→NO(g)+CO2(g) The rate constant at 701 K is measured as 2.57 M−1⋅s−1 and that at 895 K is measured as 567 M−1⋅s−1. The activation energy is 1.5×102 kJ/mol. Predict the rate constant at 525 K .

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:20, datboyjulio21
Complete the table for ion charge based upon their losing or gaining electrons in the outer shell. (use the periodic table as necessary.) group most likely ionic charge # of valence electrons i +1 ii +2 iii +3 iv +4 or -4 v -3 vi -2 vii -1 viii 0
Answers: 2

Do you know the correct answer?
The reaction between nitrogen dioxide and carbon monoxide is NO2(g)+CO(g)→NO(g)+CO2(g) The rate cons...
Questions in other subjects:

Mathematics, 10.12.2020 20:10


Geography, 10.12.2020 20:10

History, 10.12.2020 20:10



Mathematics, 10.12.2020 20:10



SAT, 10.12.2020 20:10


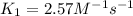 ,
, 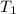 = 701 K
= 701 K
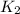 = ?
= ?
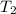 = 525 K
= 525 K
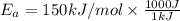
![ln[\frac{K_{2}}{K_{1}}] = (\frac{E_{a}}{R}) \times [(\frac{1}{T_{1}}) - (\frac{1}{T_{2}})]](/tpl/images/0544/3422/d5cae.png)
![ln [\frac{K_{2}}{2.57}] = \frac{150000 J/mol}{8.314 J/K mol} \times [(\frac{1}{701}) - (\frac{1}{525})]](/tpl/images/0544/3422/06744.png)
![ln [\frac{K_{2}}{2.57}]](/tpl/images/0544/3422/25ea4.png) = -8.628
= -8.628
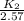 = anti ln[-8.628]
= anti ln[-8.628]
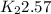 = 0.000179
= 0.000179