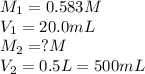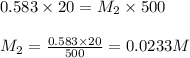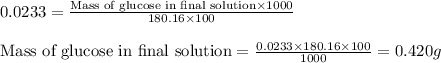A student placed 10.5 g of glucose (C6H12O6) in a volumetric fla. heggsk, added enough water to dissolve the glucose by swirling, then carefully added additional water until the 100.-mL mark on the neck of the flask was reached. The flask was then shaken until the solution was uniform. A 20.0-mL sample of this glucose solution was diluted to 0.500 L. How many grams of glucose are in 100. mL of the final solution

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:50, toniawu18
Problem page gaseous ethane reacts with gaseous oxygen gas to produce gaseous carbon dioxide and gaseous water . if of water is produced from the reaction of of ethane and of oxygen gas, calculate the percent yield of water. be sure your answer has the correct number of significant digits in it.
Answers: 2

Chemistry, 22.06.2019 09:30, strevino9178
In apex! a liquid heated beyond a certain temperature becomes
Answers: 1

Do you know the correct answer?
A student placed 10.5 g of glucose (C6H12O6) in a volumetric fla. heggsk, added enough water to diss...
Questions in other subjects:


Engineering, 17.06.2021 16:00




Mathematics, 17.06.2021 16:00


History, 17.06.2021 16:00



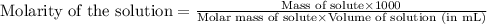 .........(1)
.........(1)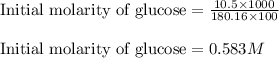
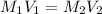
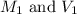 are the molarity and volume of the concentrated glucose solution
are the molarity and volume of the concentrated glucose solution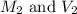 are the molarity and volume of diluted glucose solution
are the molarity and volume of diluted glucose solution