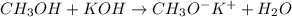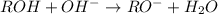Chemistry, 11.03.2020 23:19, garcikyl000
Write a reaction that shows what happens when methanol is treated with potassium hydroxide

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 07:00, haydjanggg6578
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2

Do you know the correct answer?
Write a reaction that shows what happens when methanol is treated with potassium hydroxide...
Questions in other subjects:

Business, 07.09.2021 01:10







Biology, 07.09.2021 01:10


Mathematics, 07.09.2021 01:10