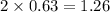At a certain temperature, the equilibrium constant, K c , for this reaction is 53.3. H 2 ( g ) + I 2 ( g ) − ⇀ ↽ − 2 HI ( g ) K c = 53.3 At this temperature, 0.800 mol H 2 and 0.800 mol I 2 were placed in a 1.00 L container to react. What concentration of HI is present at equilibrium?

Answers: 2
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 13:00, cnfndbxbfbdb2031
Which of the following are good traits of a hypothesis? it will be able to be testedit can predict an outcomeit will explain the observationsall of these
Answers: 2
Do you know the correct answer?
At a certain temperature, the equilibrium constant, K c , for this reaction is 53.3. H 2 ( g ) + I 2...
Questions in other subjects:

Social Studies, 03.11.2019 18:31

Mathematics, 03.11.2019 18:31

Biology, 03.11.2019 18:31







Physics, 03.11.2019 18:31


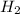 = 0.800 mole
= 0.800 mole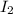 = 0.800 mole
= 0.800 mole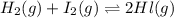
![K_c=\frac{[HI]^2}{[H_2]\times [l_2]}](/tpl/images/0543/3494/7dfaa.png)
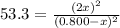
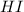 at equilibrium = 2 x =
at equilibrium = 2 x =