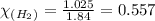Chemistry, 11.03.2020 21:28, ashl3yisbored
Consider a mixture of two gases, A and B, confined in a closed vessel. A quantity of a third gas, C, is added to the same vessel at the same temperature. How does the addition of gas C affect the following. The mole fraction of gas B?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 12:30, pleasehelpmeonthis
The difference between atomic mass and molar mass
Answers: 3

Chemistry, 22.06.2019 01:00, chrisxxxrv24
What are the variables in gay-lussac’s law? pressure and volume pressure, temperature, and volume pressure and temperature volume, temperature, and moles of gas
Answers: 1
Do you know the correct answer?
Consider a mixture of two gases, A and B, confined in a closed vessel. A quantity of a third gas, C,...
Questions in other subjects:


Mathematics, 30.03.2021 06:20


English, 30.03.2021 06:20



Physics, 30.03.2021 06:20

History, 30.03.2021 06:20


English, 30.03.2021 06:20


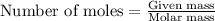 .....(1)
.....(1)