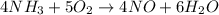Chemistry, 11.03.2020 16:45, cutebab4786
Write the reaction rate expressions for the reaction below in terms of the disappearance of the reactants and the appearance of products. Give the expressions for the disappearance of the reactants first, in the order written in the chemical equation. Then write the expressions for the appearance of the products in the order written in the chemical equation. Write the expressions in order of appearance in the equation in the form. where +- is either a plus OR a minus sign, not both, X is an integer, and A is a chemical species. 4NH3(g)+ 5O2(g) 4NO(g)+ 6H2O(g) The rate =

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 23.06.2019 00:50, alainacorkell6472
What is the enthalpy of combustion (per mole) of c4h10 (g)? –2,657.5 kj/mol –5315.0 kj/mol –509.7 kj/mol –254.8 kj/mol
Answers: 1

Do you know the correct answer?
Write the reaction rate expressions for the reaction below in terms of the disappearance of the reac...
Questions in other subjects:

History, 21.01.2020 08:31

Biology, 21.01.2020 08:31

Mathematics, 21.01.2020 08:31

Social Studies, 21.01.2020 08:31

Social Studies, 21.01.2020 08:31


Mathematics, 21.01.2020 08:31

World Languages, 21.01.2020 08:31

Biology, 21.01.2020 08:31


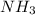 =
= ![-\frac{1d[NH_3]}{4dt}](/tpl/images/0542/8648/64c23.png)
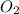 =
= ![-\frac{1d[O_2]}{5dt}](/tpl/images/0542/8648/1298a.png)
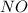 =
= ![+\frac{1d[NO]}{4dt}](/tpl/images/0542/8648/9cf34.png)
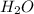 =
= ![+\frac{1d[H_2O]}{6dt}](/tpl/images/0542/8648/f3726.png)