

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:20, TamB01
Use the gizmo to find the concentration of the mystery ch3cooh. use the titrant and indicator shown below perform the titration. what is the titrant volume? titrant analyte indicator titrant volume analyte concentration naoh ch3cooh phenophthalein select one: a. 20.0 ml b. 27.0 ml c. 30.0 ml d. 24.0 ml
Answers: 2


Chemistry, 22.06.2019 21:30, shiannethorn
How many liters of 3.0 m naoh solution will react with 0.60 liters of 4.0 m h2so4? h2so4 + naoh → na2so4 + h2o 1.2 l 1.6 l 2.4 l 2.8 l
Answers: 3

Chemistry, 22.06.2019 21:50, donttrip10
What is a main difference between a mixture and a pure substance? a mixture is only a liquid, but a pure substance can be in any state. a mixture looks the same throughout, but a pure substance does not.1 a mixture can vary in composition, but a pure substance has a set composlo a mixture can be made up of a single compound, but a pure substance car
Answers: 2
Do you know the correct answer?
In the first 11.0 s s of the reaction, 1.6×10−2 mol mol of O2O2 is produced in a reaction vessel wit...
Questions in other subjects:

History, 30.01.2020 17:56

Mathematics, 30.01.2020 17:56


Chemistry, 30.01.2020 17:56


Mathematics, 30.01.2020 17:57



Social Studies, 30.01.2020 17:57

Mathematics, 30.01.2020 17:57


![\frac{d[O_2]}{dt}](/tpl/images/0542/6081/a384d.png)
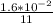 = 1.455×10⁻³ mol/second.
= 1.455×10⁻³ mol/second.



